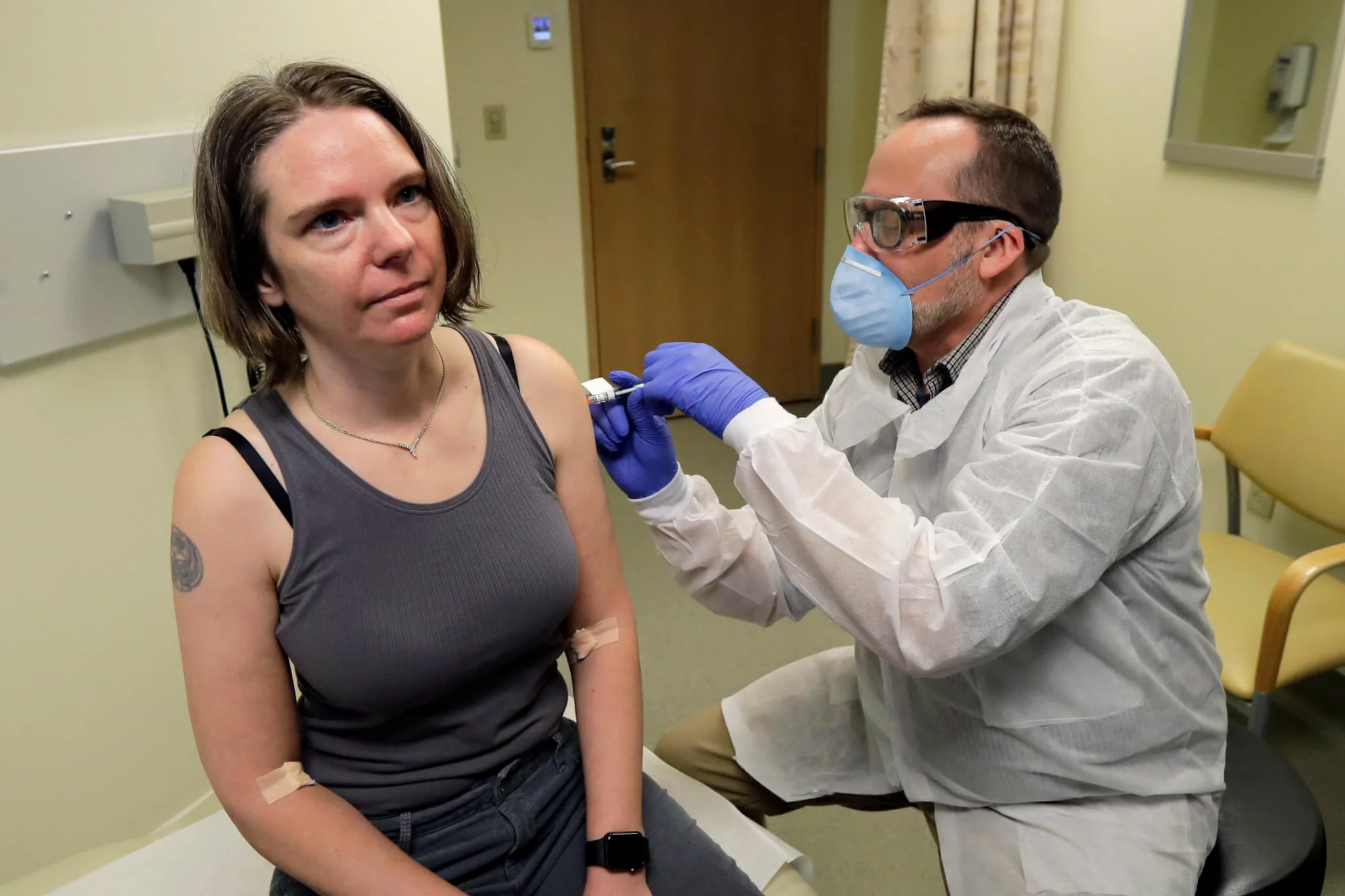
In this March 16, 2020 file photo, a pharmacist gives Jennifer Haller, left, the first shot in the first-stage safety study clinical trial of a potential vaccine for COVID-19, the disease caused by the new coronavirus, at the Kaiser Permanente Washington Health Research Institute in Seattle. The world's biggest COVID-19 vaccine test got underway July 27 with the first of 30,000 planned volunteers. (AP Photo/Ted S. Warren, File)
In the coming weeks, the US Food and Drug Administration could approve the first vaccines for COVID-19, so with that in mind, we wanted to answer some frequently asked questions about vaccines.
As 2020 comes to a close, the coronavirus pandemic is reaching its most devastating moment yet. More than 4 million people were diagnosed with COVID-19 in November, making up nearly one-third of the total number of cases in the US. Things are expected to get even worse this month, after millions of people traveled and gathered for Thanksgiving.
But there is hope. A light at the end of the tunnel: a vaccine. While America’s response to the pandemic has been nothing short of a disaster, the scientific and medical community has worked tirelessly and is on track to develop multiple effective vaccines in record time that could bring the pandemic to an end.
In the coming weeks, the US Food and Drug Administration (FDA) could approve the first vaccines for COVID-19. With that in mind, we wanted to answer some frequently asked questions about vaccines.
Is there a COVID vaccine?
There is no approved coronavirus vaccine yet, but there are dozens being tested. Researchers are currently testing 57 vaccines in clinical trials on humans, and at least 87 preclinical vaccines are under active investigation in animals, according to a New York Times database.
In the US, two vaccines in particular are close to gaining initial approval after completing clinical trials in humans. These vaccines could be available for some Americans as early as this month.
Who is making the COVID vaccine?
While there are many trials underway and companies and countries continue to work away on vaccines, the two companies behind America’s most promising vaccine trials are the pharmaceutical companies Moderna, which partnered with the National Institutes of Health, and Pfizer, which partnered with the German company BioNTech. Each company released initial data from their trials last month showing that each of their vaccines were over 90% effective in protecting people against coronavirus infection.
Both companies have applied for emergency use authorization from the FDA.
When will a COVID vaccine be available to the public?
The vaccination process will begin on a rolling basis. If the FDA signs off on Pfizer and Moderna’s COVID vaccines, some Americans could begin getting shots this month. As many as 22.5 million people could receive vaccinations before the end of the year, according to estimates from the companies.
Is the vaccine safe?
Pennsylvania’s Secretary of Health, Dr. Rachel Levine, has said she trusts the process that is giving us the vaccines.
“I feel very comfortable that there has been no politicalization of this process,” she said, “that science has driven this process.”
The vaccine development process is a rigorous, comprehensive process that begins with testing new vaccines on animals, such as mice or monkeys, to see if it produces an immune response. Scientists then give the vaccine to humans in three separate phases, beginning with a small number of people in phase one in order to test safety and dosage and determine if it triggers the immune system. In phase two, scientists give the vaccine to hundreds of people across different age ranges and at different levels of health, in order to see how the vaccine behaves among different populations. In the third and final phase, tens of thousands of people receive the vaccine, and scientists compare their health outcomes with test subjects who received a placebo. This effectively shows whether the vaccine protects against COVID-19 and whether the vaccines produce any rare or dangerous side effects.
The process is thorough and the bar to clear for FDA approval is high, incentivizing companies to take a safe, conservative approach. Multiple companies, including AstraZeneca—which conducted its trials in partnership with Oxford University—and Johnson & Johnson temporarily paused their COVID vaccine trials to investigate potentially adverse side effects in one person in each trial, before ultimately resuming them after the unexpected illnesses could not be linked to the vaccines.
As part of their applications to the FDA, both Moderna and Pfizer included two months of follow-up safety data from their Phase 3 human clinical trials that were conducted by independent bodies, such as universities. By September, Pfizer had enrolled 44,000 participants in its trial without a single serious safety issue being reported.
Some participants in the trials did experience side effects, including fever, muscle aches, headaches, and fatigue after receiving the shots, but those symptoms generally did not last more than a day. Also, such reactions are usually a sign that the body’s immune response is being activated as intended.
Who will get vaccines first?
On Tuesday, a Centers for Disease Control and Prevention (CDC) panel voted 13-1 to recommend giving America’s 21 million healthcare workers and 3 million long-term care facility residents the first coronavirus vaccine doses once they’re cleared for public use.
But even with the CDC’s decision, it will be up to state and local governments to form final vaccine plans and determine which individuals get vaccines first.
Levine said Pennsylvania health officials plan to roll out the vaccines in three phases. The first will go to the state’s front-line workers, essential workers, and vulnerable populations, like those in senior living centers.
When will other Pennsylvanians get a vaccine?
Levine said the second phase of the vaccine will cover anyone who should have gotten vaccinations in the first phase but didn’t, then it will expand to other target groups of vulnerable populations.
These groups could receive their vaccines by February and March.
The third phase will go to everyone else. Children are likely to be last in line, as the vaccines’ impact on kids has not yet been conclusively studied.
Federal health officials hope that all Americans will be able to access a vaccine by June, but that timeline could change depending on how many vaccines are ultimately authorized.
What will the vaccine cost?
Nothing. The federal government has promised that vaccines will be free to all Americans.
Where can I get the vaccine?
Once vaccines are approved, the federal government and its private partners will work with state and local governments to distribute and deliver vaccines to hospitals, doctors’ offices, health centers, pharmacies, clinics, and long-term care facilities.
Levine said Pennsylvania’s plan is for the vaccine to be distributed by health care systems and hospitals first.
Do MMR vaccines really protect you against COVID?
Recent research has shown that the Measles-Mumps-Rubella vaccine may offer some protection against being infected by the coronavirus and reduce the severity of symptoms among those diagnosed with COVID-19.
While there could be a relationship between the MMR vaccine and COVID, doctors still say that there’s not enough evidence to recommend adults get booster shots. Instead, they recommend that everyone get the COVID-19 vaccine rather than relying on the possibility that the MMR vaccine offers some protection.
Does the flu shot protect you against COVID?
No. While both are respiratory viruses, influenza and coronavirus have different properties, state health officials said, so people will need to get both vaccines.
How important is getting the flu shot this year?
Pennsylvania health officials said getting the flu shot is “essential.”
“At this point, many hospitals are running at or near capacity, and we need to get the flu vaccine as another effort to prevent our health system from being overrun,” a spokesperson for the department said.
Have Russia and China already vaccinated their citizens?
China and Russia have approved vaccines without waiting for the results of their Phase three trials and begun vaccinating some residents. Both countries are attempting to use their vaccines as an international bargaining chip with which to increase their economic and political standing in the world, but health experts say rushing the process poses serious risks.
The US is unlikely to rely on unproven Russian or Chinese vaccines, as those countries are primarily directing their stockpiles to countries with less robust medical infrastructures across Africa and the Middle East.
Will the vaccine be mandatory in the US?
It is unclear whether the federal government will attempt to mandate vaccinations. During the presidential campaign, President-elect Joe Biden said he was open to making a safe vaccine mandatory, depending on its effectiveness, the vaccine distribution process, and how rampant the virus was in the country. But he also admitted that it would be nearly impossible to actually implement a mandate.
“You can’t say, ‘Everyone has to do this,’” he said during an October town hall event.
The New York Times reported that Biden’s transition team is still discussing whether to try to institute a requirement, and state governments could also seek to pass vaccine mandates, though those would likely trigger legal battles.
Levine has repeatedly said the vaccine won’t be mandated by the state.
Employers also have the right to force their workers to be vaccinated, but workers can request exemptions based on medical reasons or religious beliefs.

For Rep. Susan Wild, supporting PA families includes reproductive rights and much more
Rep. Susan Wild wants to be very clear with Pennsylvanians: Donald Trump is committed to taking away women’s reproductive freedom, but he is not...
School districts working with anti-LGBTQ groups can cost your kids’ schools millions
Parents across South Central Pennsylvania are worried about the potential financial impacts working with anti-LGBTQ groups may have on their school...

VIDEO: Trump distances himself from his anti-abortion views
Donald Trump appeared on WGAL on Tuesday and continued to distance himself from his anti-abortion views claiming that reproductive rights are now a...

VIDEO: Community pushback gets school board to rescind decision on denying gay actor’s visit
Cumberland Valley School Board offered a public apology and voted to reinstate Maulik Pancholy as a guest speaker a week after the board voted to...

VIDEO: Project 2025 brings nuclear armageddon back into vogue
Project 2025 is a titanic document, with plans ranging from cutting half of all government employees to targeting reproductive rights on a scale...