AP Photo/John Locher
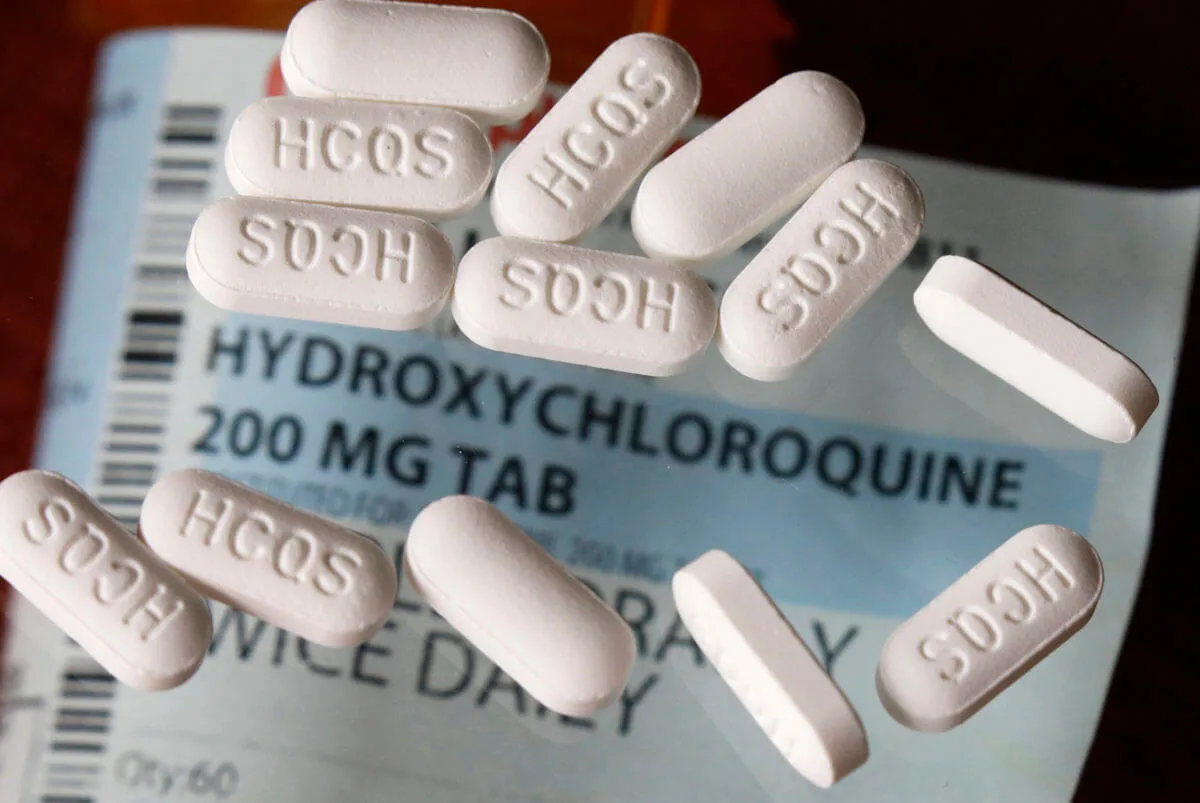
Two studies released this week found that hydroxychloroquine and chloroquine led to more fatalities among coronavirus patients.
Just one day after President Trump speculated whether injecting disinfectants into the body could treat the novel coronavirus (it can’t), the U.S. Food and Drug Administration issued a warning on Friday against Trump’s previous evidence-free, easy fix solution to COVID-19: the antimalarial drugs chloroquine and hydroxychloroquine.
In its warning, the FDA advised people not to take the drugs to treat COVID-19—despite recommendations by President Trump—unless they are in a hospital or participating in a formal clinical trial. Citing reports of “serious heart rhythm problems,” the agency cautioned against using the drugs as an outpatient and said that “Hydroxychloroquine and chloroquine have not been shown to be safe and effective for treating or preventing COVID-19.”
The drugs, also prescribed for lupus, can also cause a number of other side effects, including severely low blood pressure and muscle or nerve damage. In one report, doctors at a New York hospital said that heart rhythm abnormalities developed in most of the 84 coronavirus patients treated with hydroxychloroquine and the antibiotic azithromycin, a combo Trump has promoted.
Last month, the FDA authorized emergency use of the malaria drugs for hospitalized patients with COVID-19 who aren’t enrolled in ongoing clinical trials. But regulators said they are investigating life-threatening side effects reported with the drugs to poison control centers and other health authorities. Calls to U.S. poison control centers involving the malaria drugs have recently jumped 50%, from 52 calls in March 2019 to 79 last month, according to Dr. Christopher Hoyte of the Rocky Mountain Poison Center in Denver, Colorado.
The problems reported include irregular heart rhythms, seizures, nausea, and vomiting, Hoyte said.
The FDA’s warning comes the same day that researchers published a study in the peer-reviewed Journal of the American Medical Association that found death was a “primary outcome” of testing chloroquine as a possible treatment for COVID-19. The findings were first reported earlier this month.
Researchers tracked 81 adult patients hospitalized with COVID-19 between March 23 and April 5 at a medical facility in Brazil, before cutting the study short after some patients developed irregular heart rates and nearly two dozen died after taking daily doses of the drug. Heart abnormalities were also detected in 11 of the total 81 patients.
“One can only conclude from this trial that high-dose chloroquine (and by close association, hydroxychloroquine) in combination and azithromycin and possibly oseltamivir, is potentially associated with increased mortality among patients with severe, suspected COVID-19,” wrote Stephan Fihn, a physician at Harborview Medical Center in Washington state, in a JAMA editorial published with the study’s results.
The Brazilian study’s findings are similar to those of a study conducted in United States veterans hospitals, which found that there were more deaths among those given hydroxychloroquine versus standard care. In that study, which was released earlier this week, about 28% of patients who were given hydroxychloroquine plus usual care died, versus only 11% of those getting routine care alone.
Despite the findings of that study, Veterans Affairs Secretary Robert Wilkie has continued to promote hydroxychloroquine, claiming without evidence that it’s safe and that it has been effective for young and middle-aged veterans in particular.
RELATED: Trump Called Hydroxychloroquine a ‘Game Changer.’ A Study Found It Led to More Deaths.
“That’s an observational study. It’s not a clinical study,” Wilkie said during an interview with MSNBC on Wednesday. “It was done on a small number of veterans. Sadly, those of whom were in the last stages of life. And the drug was given to them. And I have to also say that the drug — we know the drug has been working on middle-age and younger veterans … working in stopping the progression of the disease.”
An Associated Press fact check of Wilkie’s statements found that he mischaracterized the study’s findings as he “delved into an area of speculation by amateurs that his own agency” called “inaccurate and irresponsible.”
“Wilkie rejected a study that relies on his department’s own hospital data and finds no benefit from hydroxychloroquine,” the AP found. “His claim that it helps younger or middle-aged veterans with COVID-19 is also unsubstantiated.”
There are no proven treatments for COVID-19, but citing two scientifically-questionable studies, Trump has for more than a month loudly touted chloroquine and hydroxychloroquine as a possible “game changer” in the fight against coronavirus. While there had been some initial indications the drug may prove effective in fighting COVID-19, leading public health experts— including Dr. Anthony Fauci, the nation’s top infectious disease doctor and member of Trump’s coronavirus task force—repeatedly cautioned against believing the drug was a sure-fire treatment for coronavirus and emphasized the need for clinical trials.
“We still need to do the definitive studies to determine whether any intervention, not just this one, is truly safe and effective,” Fauci, the director of the National Institute of Allergy and Infectious Diseases, said on Fox News earlier this month.
Trump ignored the advice of Fauci and others and his reckless promotion of the drugs spread like wildfire on social media. From there, the hype surrounding the drugs quickly devolved into widespread misinformation, causing a shortage as Americans, including doctors and pharmacists, embraced the possibility of chloroquine and hydroxychloroquine as a treatment for COVID-19 and began hoarding the drugs.
RELATED: Doctors Are Hoarding Drugs Lupus Patients Need Because They Might Treat Coronavirus
Having seen Trump tout the drugs on television, one Arizona man even decided to take what he thought was chloroquine to guard against contracting coronavirus. But the man ingested the wrong formulation of chloroquine—which can also be developed into an aquarium cleaner—and died within hours.
Trump’s embrace of the drug was driven by a desire to get a “win,” and political appointees in his administration, eager to please the president, exerted massive pressure on career health officials to try to deliver that “win.” According to documents obtained by Vanity Fair, on April 4, Brett Giroir, the assistant secretary for health in the Department of Health and Human Services, sent an email with the subject line “Hydroxychloroquine” to a group including FEMA administrator Pete Gaynor, HHS assistant secretary for preparedness and response Robert Kadlec, and Navy Rear Admiral John Polowczyk, who leads a supply-chain task force at FEMA.
In his email, Giroir wrote that officials at the White House “really want to flood Ny and NJ with treatment courses” of the drug. New York and New Jersey, of course, have been the hardest hit states in the nation by the coronavirus pandemic and together comprise 42% of the nation’s confirmed COVID-19 cases and more than half of its deaths.
Giroir wondered if they should send “a few million” of the federal government’s 29 million pill stockpile of the drug, which it had amassed after Trump’s embrace of the unproven drug.
On that same day, April 4, Trump had once again publicly embraced hydroxychloroquine. “I’ll say it again: What do you have to lose?” Mr. Trump said during his daily press conference. “Take it.”
While Giroir and other political appointees raced to try and please Trump, one administration official dared to push back and ask for more serious vetting of hydroxychloroquine. Dr. Rick Bright, the federal official who led the Biomedical Advanced Research and Development Authority (BARDA), the federal agency involved in developing a coronavirus vaccine, cautioned against the drug, only to then lose his job earlier this week.
In a statement to the New York Times on Wednesday, Barda said he was demoted for resisting efforts “to fund potentially dangerous drugs” and that the administration had put “politics and cronyism ahead of science.”
RELATED: 4 People Who’ve Been Punished After Criticizing Trump’s Coronavirus Response
“Specifically, and contrary to misguided directives, I limited the broad use of chloroquine and hydroxychloroquine, promoted by the administration as a panacea, but which clearly lack scientific merit,” Bright said. “While I am prepared to look at all options and to think ‘outside the box’ for effective treatments, I rightly resisted efforts to provide an unproven drug on demand to the American public.”
The president, however, continues to speculate about unproven—if not dangerous and potentially deadly—treatments for the coronavirus.
On Friday, as backlash raged over Trump’s baseless suggestion that disinfectants might kill COVID-19, White House press secretary Kayleigh McEnany told the Washington Post that Trump believes Americans should consult with doctors regarding coronavirus treatments.
Additional reporting from the Associated Press.

For Rep. Susan Wild, supporting PA families includes reproductive rights and much more
Rep. Susan Wild wants to be very clear with Pennsylvanians: Donald Trump is committed to taking away women’s reproductive freedom, but he is not...
School districts working with anti-LGBTQ groups can cost your kids’ schools millions
Parents across South Central Pennsylvania are worried about the potential financial impacts working with anti-LGBTQ groups may have on their school...

VIDEO: Trump distances himself from his anti-abortion views
Donald Trump appeared on WGAL on Tuesday and continued to distance himself from his anti-abortion views claiming that reproductive rights are now a...

VIDEO: Community pushback gets school board to rescind decision on denying gay actor’s visit
Cumberland Valley School Board offered a public apology and voted to reinstate Maulik Pancholy as a guest speaker a week after the board voted to...

VIDEO: Project 2025 brings nuclear armageddon back into vogue
Project 2025 is a titanic document, with plans ranging from cutting half of all government employees to targeting reproductive rights on a scale...